- Stock: In Stock
- Brand: Incepta Pharmaceuticals
- Product ID: Ciprofloxacin
100% Secure Payment

This Item is for pre order
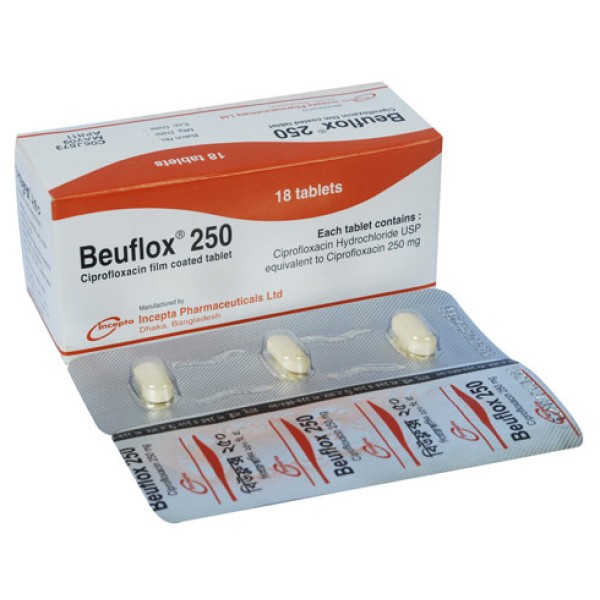
Beuflox 250
Indications
Ciprofloxacin is indicated for the treatment of the following infections caused by sensitive bacteria:
Severe systemic infections: e.g; septicemia, bacteremia, peritonitis, infections in immunosuppressed patients with haematological or solid tumors and in patients in intensive care unit with specific problems such as infected burns.
Respiratory tract infections: Lobar and broncho pneumonia, acute and chronic bronchitis and empyema.
Urinary tract infections: Uncomplicated and complicated urethritis, cystitis, pyelonephritis, prostatitis and epididymitis.
Skin and soft tissue infections: Infected ulcers, wound infections, abscesses, cellulitis, otitis externa, erysipelas and infected burns.
Gastrointestinal infections: Enteric fever, infective diarrhea.
Infections of the biliary tract: Cholangitis, cholecystitis, empyema of the gall bladder.
Intra-abdominal infections: Peritonitis, intra abdominal abscesses.
Bone and joint infections: Osteomyelitis, septic arthritis.
Pelvic infections: Salpingitis, endometritis, pelvic inflammatory diseases.
Eye, ear, nose and throat infections: Otitis media, sinusitis, mastoiditis, tonsillitis.
Gonorrhoea: Urethral, rectal and pharyngeal gonorrhoea caused by beta-lactamase producing organism or organisms moderately sensitive to penicillin.
Dosage & Administration
Adult Dose:
For oral dosage &suspension:
Urinary Tract infection: Acute uncomplicated: 250 mg twice daily for 3 days; Mild/Moderate: 250 mg twice daily for 7 to 14 days; Severe/Complicated: 500 mg twice daily for 7 to 14 days; Chronic Bacterial Prostitis : 500 mg twice daily for 28 days; Lower Respiratory Tract infection: Mild/Moderate: 500 mg twice daily for 7 to 14 days, Severe/Complicated : 750 mg twice daily for 7 to 14 days; Acute Sinusitis : 500 mg twice daily for 10 days; Skin and Skin Structure infection: Mild/Moderate : 500 mg twice daily for 7 to 14 days, Severe/Complicated : 750 mg twice daily for 7 to 14 days, Bone and joint infection: Mild/Moderate 500 mg twice daily for 4 to 6 weeks, Severe/Complicated : 750 mg twice daily for 4 to 6 weeks, Intra Abdominal Infection: 500 mg twice daily for 7 to 14 days, Infectious Diarrhea: Mild/Moderate/Severe: 500 mg twice daily for 5 to 7 days, Typhoid Fever : 500 mg twice daily for 10 days, Urethral & Cervical Gonococcal Infections: Uncomplicated: 250 mg Single dose.
For IV infusion :
Urinary Tract Infection: Mild to Moderate: 200 mg 12 hourly for 7-14 days;Severe or Complicated: 400 mg 12 hourly for 7-14 days; Lower Respiratory Tract infection: Mild to Moderate: 400 mg 12 hourly for 7-14 days; Severe or Complicated: 400 mg 8 hourly for 7-14 days; Nosocomial Pneumonia: Mild/Moderate/Severe: 400 mg 8 hourly for 10-14 days; Skin and Skin Structure: Mild to Moderate: 400 mg 12 hourly for 7-14 days; Severe or Complicated: 400 mg 8 hourly for 7-14 days; Bone and Joint Infection: Mild to Moderate: 400 mg 12 hourly for more than 4-6 weeks; Severe/Comlicated: 400 mg 8 hourly for more than 4-6weeks; Intra abdominal (Acute abdomen): Complicated: 400 mg 12 hourly for 7-14 days; Acute Sinusitis: Mild/Moderate: 400 mg 12 hourly for10 days: Chronic Bacterial Prostatitis: Mild/Moderate: 400 mg 12 hourly for 28 Days.
Children and adolescents:
RTI & GI infections: Neonate-15mg/kg twice daily, Child (1 month -18 years)-20mg/kg (max 750 mg) twice daily; UTI: Neonate-10 mg/kg twice daily, Child (1 month -18 years)-10mg/kg (max 750 mg) twice daily; Pseudomonal lower respiratory tract infection in cystic fibrosis: Child (1 month -18 years) - 20mg/kg (max 750 mg) twice daily; Anthrax (treatment & post exposure prophylaxis): Child (1 month -18 years) - 20mg/kg (max 750 mg) twice daily.
Use in Pregnancy and Lactation
Reproduction studies performed in rats and rabbits using parenteral and oral administration did not reveal any evidence of teratogenicity, impairment of fertility or impairment of pre or postnatal development. However, as with other quinolones, Ciprofloxacin has been shown to cause arthropathy in immature animals and therefore, its use during pregnancy is not recommended. Studies in rats have indicated that Ciprofloxacin is secreted in milk, administration to nursing mothers is thus not recommended.
Side Effects
Ciprofloxacin is generally well tolerated. Frequent adverse reactions are- Gastrointestinal disturbance: e.g. nausea diarrhea, vomiting, dyspepsia, abdominal pain. Disturbance of the CNS: e.g. dizziness, headache, tiredness, confusion, convulsions. Hypersensitivity reactions: e.g. skin rashes, pruritus, and possible systemic reactions. Other possible side effects are - joint pain, light sensitivity, transient increase in liver enzyme (especially in patients with history of liver damage), serum bilirubin, urea or serum creatinine. Arthralgia and myalgia may also occur.
Precautions
Ciprofloxacin should be used with caution in patients with a history of convulsive disorders. Crystalluria related to the use of Ciprofloxacin has been observed only rarely. Patients receiving Ciprofloxacin should be well hydrated to avoid excessive alkalinity of the urine.
Beuflox Injection should only be administered by slow intravenous infusion over a period of 60 minutes. Local IV site reactions have been reported with the intravenous administration of Ciprofloxacin. These reactions are more frequent if infusion time is 30 minutes or less or if small vein of the hand are used.
Use in Pregnancy & Lactation
Reproduction studies performed in rats and rabbits using parenteral and oral administration did not reveal any evidence of teratogenicity, impairment of fertility or impairment of pre or postnatal development. However, as with other quinolones, Ciprofloxacin has been shown to cause arthropathy in immature animals and therefore, its use during pregnancy is not recommended. Studies in rats have indicated that Ciprofloxacin is secreted in milk, administration to nursing mothers is thus not recommended.
Drug Interaction
Concurrent administration of Ciprofloxacin with theophylline may lead to elevated plasma concentrations of theophylline. Plasma level of theophylline should be monitored and dosage adjustments made as appropriate. Antacid containing magnesium hydroxide or aluminium hydroxide may interfere with the absorption of Ciprofloxacin & concurrent administration of these agents with Ciprofloxacin should be avoided. Probenecid interferes with renal tubular secretion of Ciprofloxacin and produces an increase in the level of Ciprofloxacin in the serum. As with other broad spectrum antibiotics prolonged use of Ciprofloxacin may result in over growth of non-susceptible organisms. Repeated evaluation of patient\\\\\\\'s conditions and microbial susceptibility testing is essential. If superinfections occur during therapy, appropriate measure should be taken.
Over Dose
In case of acute overdose, the patient should be carefully observed and given supporative treatment, including monitoring of renal function. Adequate hydration must be maintained.























%20Pvt.%20Ltd./Movicol-Oral-Powder-190x190.jpg)
